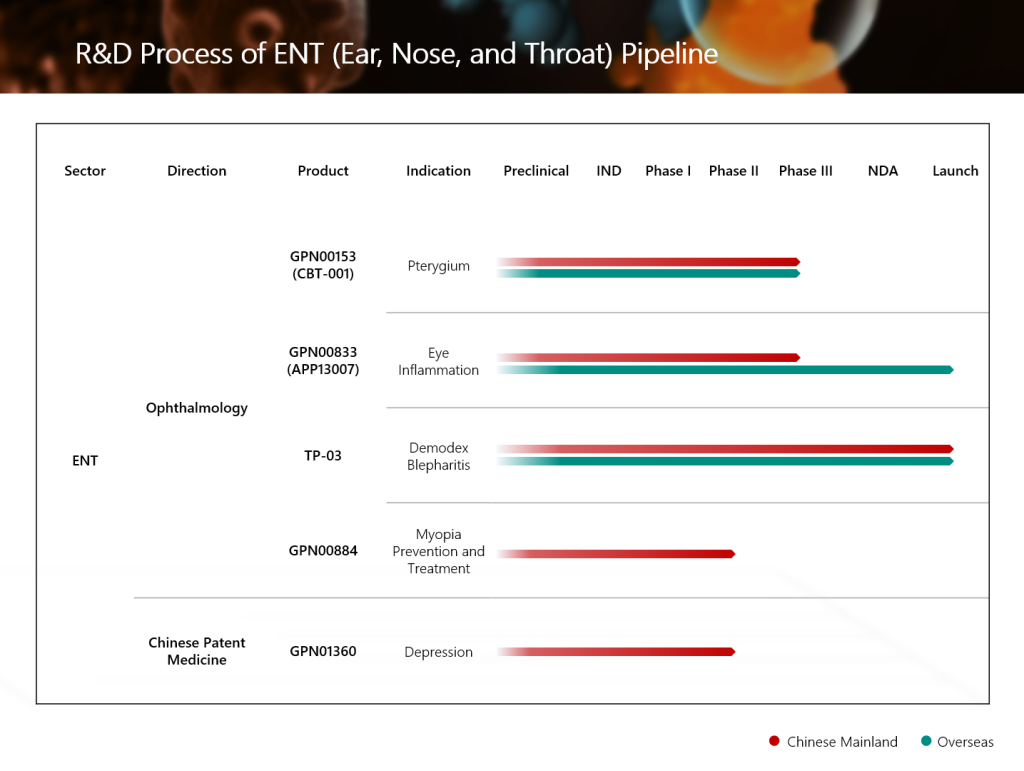
The global innovative ophthalmic product GPN01768 [TP-03, (lotilaner ophthalmic solution) 0.25%] introduced by Grand Pharma for the treatment of Demodex blepharitis has been granted Drug Registration Certificate by the National Medical Products Administration of the People’s Republic of China recently. The approval was granted with no supplementary filing requirements during the drug review process, achieving a “zero supplementation request” (零發補) approval.
In March 2024, Grand Pharma entered into a strategic cooperation agreement for product introduction with Tarsus Pharmaceuticals, Inc. (“Tarsus Pharmaceuticals”, the developer of TP-03), and acquired the exclusive development, manufacture and commercialization rights in Greater China Region (Mainland China, Hong Kong Special Administrative Region of China, Macao Special Administrative Region of China, and Taiwan Region of China). The approval of the product’s commercialization marks another significant advancement for Grand Pharma in the direction of innovative ophthalmic drugs in the ENT sector.
GPN01768 was approved for commercialization by the United States Food and Drug Administration in July 2023, becoming the first and only product approved for the treatment of Demodex blepharitis globally.
GPN01768 generated revenue of approximately USD180 million in the United States in 2024, and generated revenue of approximately USD450 million in the United States in 2025.
GPN01768 was approved for commercialization in Macao Special Administrative Region of China in May 2025. Grand Pharma will then actively promote the registration and application process for the product in Hong Kong Special Administrative Region of China and Taiwan Region of China.