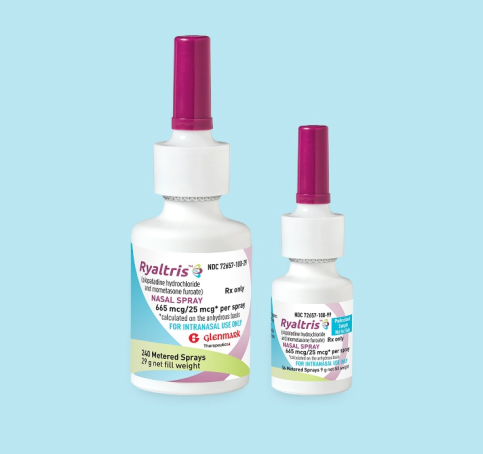
The New Drug Application (NDA) of Ryaltris® Compound Nasal Spray (GSP 301 NS), Grand Pharma’s global innovative frug for the treatment of allergic rhinitis (AR) in adults and children, that submitted to China National Medical Products Administration (NMPA) has been accepted recently. This is another important R&D progress of the Group in the field of respiratory and severe disease anti-infection.
GSP 301 NS is a novel antihistamine and corticosteroid combination nasal spray. It has been approved for commercialization in many countries and regions, including the United States, Australia, Russia, South Korea, the United Kingdom and the European Union.
Clinical results showed that the efficacy ratings of GSP 301 NS are better than the monomer originator preparations Patanase® NS and Nesunar® NS. The safety, tolerability and pharmacokinetic profile of GSP 301 NS also met the preset clinical endpoints.
According to the “Allergic Rhinitis and Its Impact on Asthma (ARIA)” guidelines, nasal antihistamines and nasal corticosteroids are the preferred medications for AR. For patients with moderate to severe AR, it is recommended to use the combination of nasal antihistamines and nasal corticosteroids.